The similar lattice constants for alkali and alkaline earth fluorides versus hydrides facilitates the formation of solid solutions, which have been considered for hydrogen storage applications. 
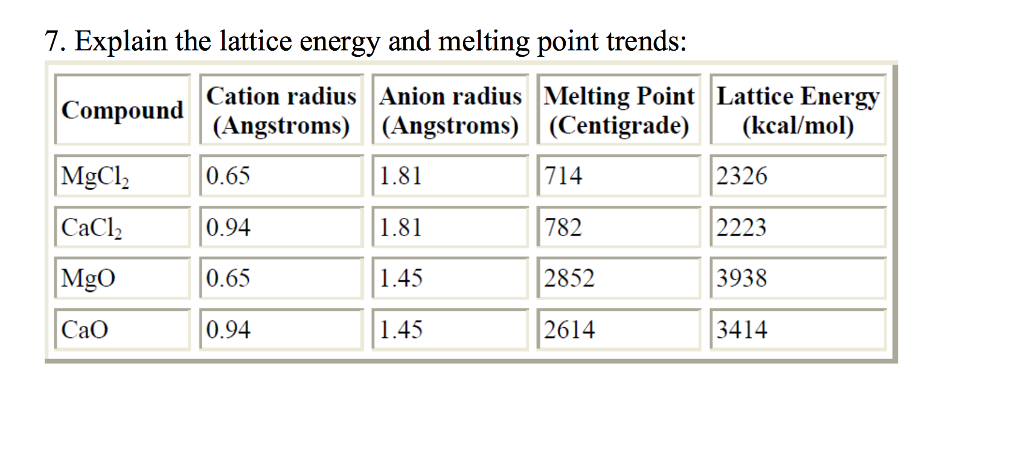
Essentially, the hydride ion holds its valence electrons so loosely that they wander over slightly more volume than the greater number of, but more tightly held, electrons in fluoride ions. We would expect that for the same ion charges and crystal structure (the latter true of all the compounds cited above), we would expect smaller lattice constant to correspond to more lattice energy, and this we see. First, we can see that by increasing the charge of the ions, we will dramatically increase the lattice energy. Lattice constants in Angstroms (Å) are from the Wikipedia articles on the respective compounds, lattice energies in kJ/mol are quoted from. Lattice Energy Trend in Periodic Table From the above tables, one can observe that the lattice energy increases with atomic charge and decreases with ionic radius. Lattice energy trend Looking at the Kapustinskii equation above, we can begin to understand some of the lattice energy trends as we move across and down the periodic table. Where the crystal structure is the same we can assess the distance from Lattice constants, which are themselves a little squishy but at least accessible by direct (X-ray diffraction) experiments. Jump to navigationJump to search Lattice enthalpy of an ionic solid is defined as the energy required to completely separate one mole of a solid ionic compound into gaseous constituent. We should be looking at distances between ion centers and crystal structures. Everything in ordinary chemistry is at least somewhat squishy. This definition causes the value for the lattice energy to always be positive, since this will always be an endothermic reaction. In one definition, the lattice energy is the energy required to break apart an ionic solid and convert its component atoms into gaseous ions. As suggested in the comments, hydride ion size is founded on shifting sands, and even the size of the cation is not really fixed. Lattice Energy is a type of potential energy that may be defined in two ways. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
